- Your cart is empty
- Continue Shopping

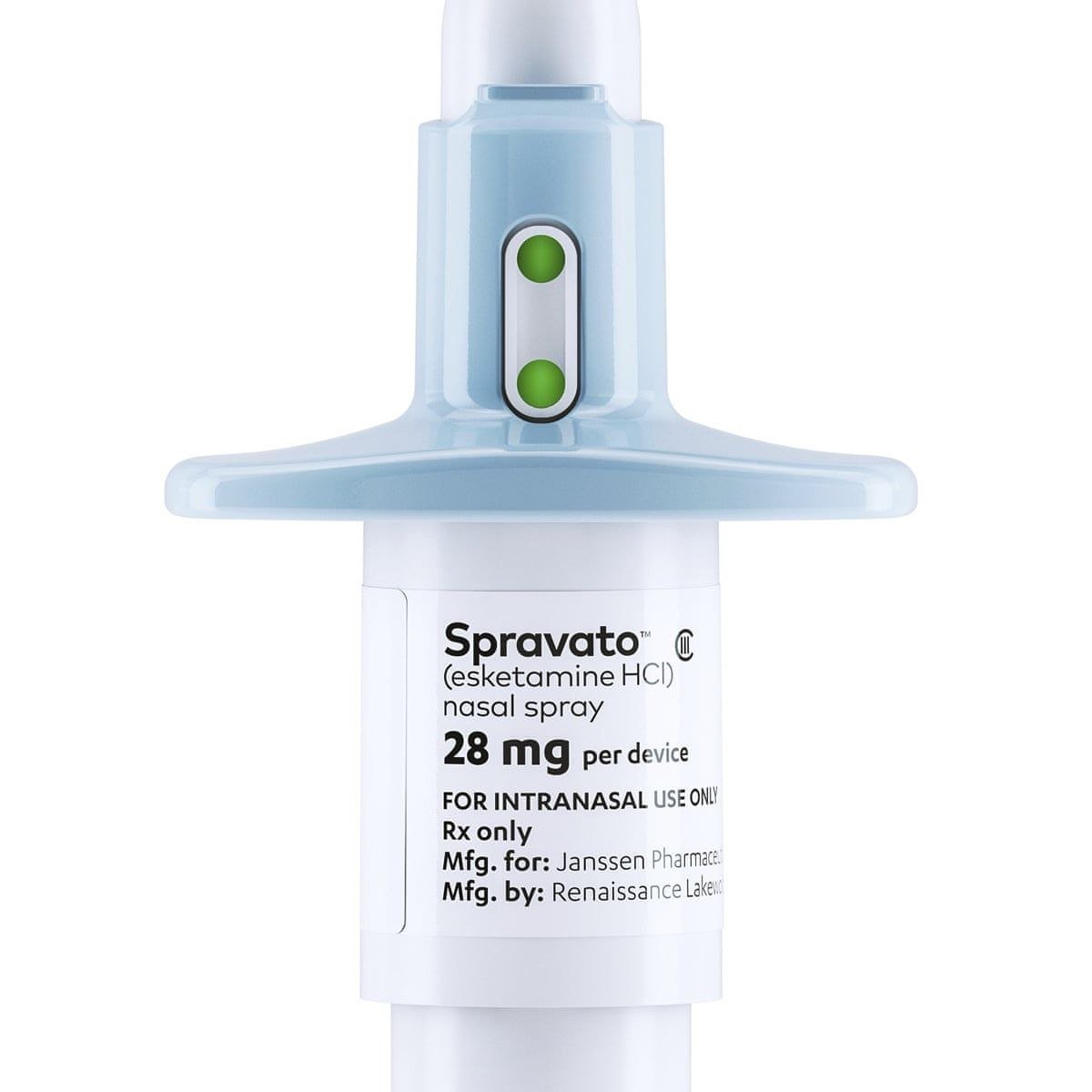
Esketamine Nasal Spray
€160 – €550Price range: €160 through €550
Product Description
Esketamine Nasal Spray
Esketamine nasal spray is a prescription medication approved by the FDA in 2019 for treating treatment-resistant depression (TRD) in adults, and later expanded to include depression with acute suicidal ideation or behavior (in combination with an oral antidepressant). It’s the (S)-enantiomer of ketamine, a dissociative anesthetic, but formulated at a lower dose for psychiatric use. Unlike traditional antidepressants (e.g., SSRIs), it acts rapidly—often within hours—and targets the glutamate system in the brain via NMDA receptor antagonism.
Key Uses
- Primary indication: TRD (depression not responding to ≥2 prior antidepressants).
- Secondary: Major depressive disorder (MDD) with imminent suicide risk.
- Administered only in certified healthcare settings under the SPRAVATO REMS program (Risk Evaluation and Mitigation Strategy) due to risks like sedation, dissociation, and potential abuse.
How It Works
Esketamine blocks NMDA receptors, increasing glutamate release and activating AMPA receptors. This promotes synaptogenesis (new neural connections) in brain areas like the prefrontal cortex, disrupted in depression. Effects peak in 40 minutes, lasting days to weeks.
Evidence:
- TRANSFORM trials (Phase 3): Esketamine + oral antidepressant showed 70% response rate vs. 52% placebo at week 4 (MADRS score reduction ≥50%).
- SUSTAIN trials: Sustained benefits over 1 year in maintenance phase.
- Meta-analyses (e.g., JAMA Psychiatry 2020) confirm rapid antidepressant effects, superior to placebo but with higher dropout due to side effects.
Dosage and Administration
| Dose | Schedule | Notes |
|---|---|---|
| Induction**: 56 mg or 84 mg (2-3 sprays per nostril) | Twice weekly for 4 weeks | Supervised; 2-hour monitoring post-dose. |
| Induction**: Same doses | Weeks 5-8: Once weekly | Taper based on response. |
| Maintenance**: 56-84 mg | Every 1-2 weeks | Long-term as needed. |
- Self-administration prohibited; must be under medical supervision.
- Avoid food 2 hours before; no alcohol/caffeine.
Side Effects
Common (≥20% patients):
- Dissociation (e.g., feeling detached, 40-75%).
- Dizziness/vertigo.
- Nausea, headache.
Serious (black box warnings):
- Sedation/abuse potential: Respiratory depression risk; Schedule III controlled substance.
- Blood pressure spikes (monitor 2 hours post-dose).
- Suicidality risk in young adults (monitor closely).
- Rare: Mania, cognitive impairment.
Evidence: In trials, 5% discontinued due to dissociation; BP increases >90 mmHg systolic in 10-20%.
Contraindications and Warnings
- Avoid with: MAOIs (14-day washout), psychostimulants, CNS depressants.
- Not for: Aneurysms, arteriovenous malformations, recent heart attack/stroke.
- Pregnancy: Category not assigned; limited data—avoid (potential fetal harm).
- Driving: No operation of vehicles/machinery until next day.
Comparisons
| Treatment | Onset | Efficacy (TRD) | Administration | Cost (US, approx.) |
|---|---|---|---|---|
| Esketamine | Hours-Days | 60-70% response | Clinic, nasal | $600-900/dose |
| IV Ketamine** (off-label) | Hours | Similar | Clinic, IV | $400-800/session |
| SSRIs/SNRIs | 4-6 weeks | 40-50% | Home, oral | $10-50/month |
| ECT | Days | 70-90% | Clinic, procedure | $2,000+/session |
Access and Cost
- US: ~1,000 certified centers; insurance often covers for TRD (e.g., Medicare Part B).
- Global: Approved in EU, Canada, Australia; limited elsewhere.
- Generic: None yet (patent until ~2033).
Sources: FDA label (Spravato.com), APA guidelines, New England Journal of Medicine reviews (2019-2023), clinicaltrials.gov (NCT02417064, etc.). Always consult a psychiatrist—not a substitute for medical advice. If you’re considering it, discuss with a provider experienced in ketamine therapies.
Additional Information
| Quantity | 10 Bottles, 25 Bootles, 50 Bottles |
|---|
Related products
Botox 100 Unit
€280 – €2,500Price range: €280 through €2,500Gouri
€114