- Your cart is empty
- Continue Shopping

Product Description
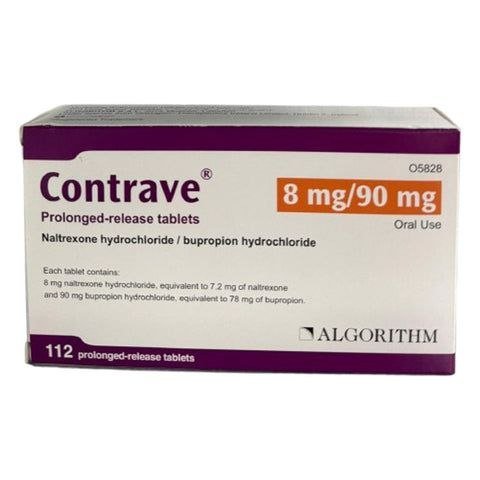
Contrave Tablets
Contrave tablets (naltrexone HCl and bupropion HCl extended-release tablets) is a prescription medication approved by the FDA in 2014 for chronic weight management in adults with obesity (BMI ≥30 kg/m²) or overweight (BMI ≥27 kg/m²) who have at least one weight-related comorbidity, such as type 2 diabetes, hypertension, or dyslipidemia. It is used as an adjunct to a reduced-calorie diet and increased physical activity. It is not intended for cosmetic weight loss or short-term use.
How It Works
Contrave combines two active ingredients:
- Naltrexone HCl (8 mg per tablet): An opioid receptor antagonist that may reduce appetite and food cravings by modulating reward pathways in the brain (e.g., blocking the hedonic effects of food).
- Bupropion HCl (90 mg per tablet): A norepinephrine-dopamine reuptake inhibitor (NDRI) antidepressant that helps suppress appetite and may increase energy expenditure.
The extended-release formulation provides 24-hour coverage. Typical dosing starts at 1 tablet (8/90 mg) once daily for Week 1, escalating to a maximum of 2 tablets twice daily (total 32/360 mg/day) by Week 4, if tolerated.
Evidence: Clinical trials (e.g., COR-I and COR-II studies) showed ~5% average weight loss over 56 weeks vs. placebo (4-5% responders achieving ≥5% loss). A 2020 meta-analysis in Obesity Reviews confirmed modest efficacy (3-5 kg more than placebo) but emphasized lifestyle integration.
Dosage Forms
- Strength: 8 mg naltrexone / 90 mg bupropion per oblong, film-coated tablet (yellow; imprinted “8/90”).
- Packaging: Bottles of 60 or 120 tablets (28-day or 56-day supply at max dose).
- Administration: Swallow whole with water; take with meals to reduce nausea. Avoid crushing/chewing.
Common Side Effects
| Category | Frequent (>5%) | Serious (Rare) |
|---|---|---|
| GI | Nausea (32-36%), constipation (19%), vomiting (10-11%), diarrhea (7-9%) |
|
| Neuropsych | Headache (18%), dizziness (9-10%), insomnia (9%), dry mouth (8%) | Seizures (0.1%; risk ↑ with dose/history), suicidal thoughts (black box warning) |
| CV | Increased heart rate (↑5-9 bpm), hypertension | Increased BP/HR |
| Other | Hyperhidrosis (sweating, 6-7%) | Glaucoma (angle-closure), allergic reactions, hepatotoxicity |
Discontinuation: Taper if stopping bupropion to avoid withdrawal; naltrexone has no taper needed.
Contraindications & Warnings
- Absolute: Uncontrolled hypertension, seizures/epilepsy, bulimia/anorexia history, chronic opioid use (precipitates withdrawal), MAOI use (within 14 days), pregnancy (Category X).
- Precautions: Monitor BP/HR; avoid alcohol (seizure risk); not for smokers quitting (use Zyban instead). Hepatic/renal impairment requires dose adjustment.
- Drug Interactions: Strong CYP2B6 inhibitors (e.g., ticlopidine) ↑ bupropion levels; opioids/dextromethorphan blocked by naltrexone.
Availability & Cost
- Brand-only (no generic as of 2024).
- US retail: ~1,300/month without insurance; copay cards from manufacturer (Currax) can reduce to 99.
- Covered by many insurers for eligible patients.
Important: This is general information based on FDA labeling, prescribing info (currax.com), and studies (e.g., NEJM 2010; JAMA 2015). Consult a healthcare provider for personalized advice, as Contrave requires a Risk Evaluation and Mitigation Strategy (REMS) program enrollment due to seizure risk. Do not self-medicate. If experiencing side effects, seek medical help immediately.