- Ihr Warenkorb ist leer
- Weiter Einkaufen

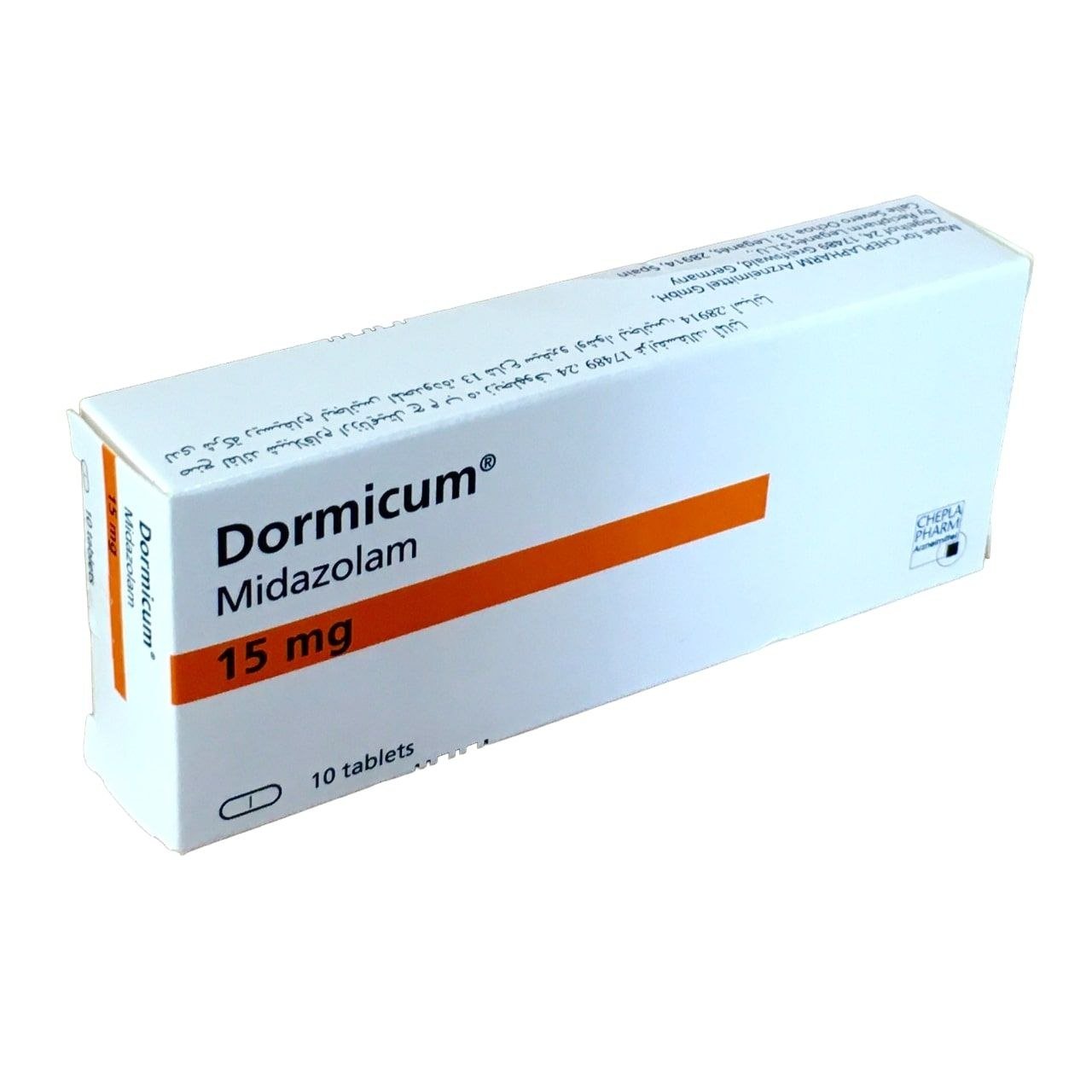
Midazolam Dormicum
€75
Add to cart
Jetzt Kaufen
Kategorie: Angst PillenSchlafmittel
Tags: Midazolam Dormicum
Produkt Beschreibung
Midazolam Dormicum
Midazolam Dormicum, commonly sold under the brand name Dormicum, is a short-acting benzodiazepine medication used primarily as a sedative, anxiolytic (anti-anxiety), muscle relaxant, and anticonvulsant. It’s a Schedule IV controlled substance in many countries due to its potential for abuse and dependence. Here’s a factual breakdown:
Key Uses (Approved Indications)
- Sedation and Anesthesia: Pre-operative sedation, conscious sedation during procedures (e.g., endoscopy, dental work), and induction/maintenance of anesthesia in surgery.
- Seizure Control: Treatment of status epilepticus (prolonged seizures), often via IV or intranasal routes.
- Anxiolysis: Short-term relief of severe anxiety or agitation in hospital settings.
- Andere: ICU sedation for mechanically ventilated patients; sometimes off-label for insomnia or procedural anxiety.
Pharmacology and Mechanism
- Klasse: Imidazobenzodiazepine (a subtype of benzodiazepines).
- So funktioniert es: Enhances the effect of GABA (gamma-aminobutyric acid), the brain’s main inhibitory neurotransmitter, leading to sedation, amnesia, reduced anxiety, and anticonvulsant effects.
- Onset and Duration:
| Route | Beginn | Dauer |
|---|---|---|
| IV/IM | 1-5 minutes | 1-6 hours |
| Oral (syrup/tablet) | 15-30 minutes | 2-6 hours |
| Intranasal | 5-10 minutes | 1-4 hours |
- Halbwertszeit: 1.5-2.5 hours in adults (longer in elderly/children/liver impairment).
Dosage Forms and Typical Doses (Adults; Consult a Doctor)
- Injectable: 1 mg/mL, 5 mg/mL solutions.
- Oral: Syrup (2 mg/mL), tablets (7.5 mg, 15 mg as Dormicum).
- Examples:
- Pre-op sedation: 0.07-0.08 mg/kg IV.
- Seizures: 0.2 mg/kg IV (max 10 mg).
- Oral anxiety: 7.5-15 mg.
Nebenwirkungen und Risiken
| Häufig (Milde) | Ernst (Suchen Sie Sofort Hilfe) |
|---|---|
| Drowsiness, dizziness | Respiratory depression, apnea |
| Headache, nausea | Hypotension, cardiac arrest |
| Amnesia, confusion | Paradoxical agitation, coma |
| Ataxia (impaired coordination) | Dependence/withdrawal (seizures, tremors) |
- Überdosis: Can cause profound sedation, respiratory failure. Treated with flumazenil (benzodiazepine antagonist).
- Kontraindikationen: Acute narrow-angle glaucoma, severe respiratory insufficiency, hypersensitivity. Use caution with alcohol, opioids, or other CNS depressants (risk of fatal overdose).
- Widerrufsrecht: Abrupt stopping after prolonged use can cause rebound anxiety, insomnia, seizures—taper gradually.
Evidence and Facts
- Wirksamkeit: Supported by RCTs and guidelines (e.g., American Society of Anesthesiologists for procedural sedation; FDA-approved since 1985).
- Verfügbarkeit: Prescription-only worldwide. Dormicum is a Roche brand, common in Europe, Asia, Latin America (e.g., 15 mg tablets).
- Rechtlicher Status: Controlled (e.g., DEA Schedule IV in US; similar elsewhere). Not for recreational use—high abuse potential leads to black-market diversion.
- Studien: Meta-analyses (e.g., Cochrane reviews) confirm superior short-acting profile vs. longer benzodiazepines like diazepam for procedures.
Wichtig: This is general information from medical sources (e.g., FDA labels, Lexicomp, PubMed). Nehmen Sie keine Selbstmedikation vor.. Always consult a healthcare professional for personalized advice, as misuse can be life-threatening. If you’re experiencing a medical emergency, call emergency services.
Verwandte Produkte
Wählen Sie Optionen
Dieses Produkt hat mehrere Varianten. Die Optionen können gewählt werden auf der Produkt-Seite