- Your cart is empty
- Continue Shopping
Product Description
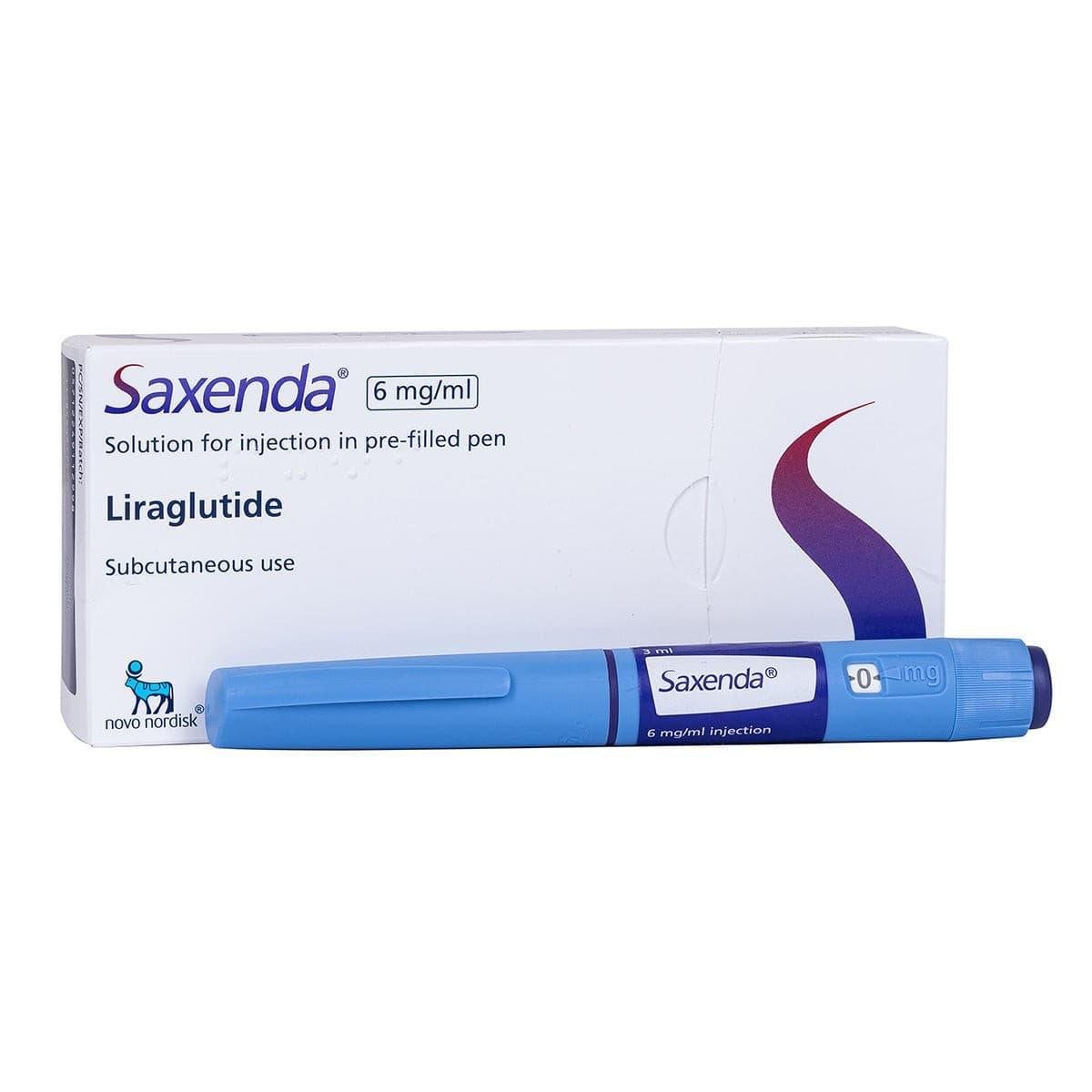
Saxenda Liraglutide
Saxenda is a brand-name prescription medication containing liraglutide (3 mg/3 mL solution for subcutaneous injection). It’s a GLP-1 receptor agonist (glucagon-like peptide-1), mimicking a hormone that regulates appetite and blood sugar. Developed by Novo Nordisk, it’s FDA-approved specifically for chronic weight management in adults and children aged 12+ with obesity or overweight conditions with related health issues (e.g., hypertension, type 2 diabetes, dyslipidemia).
Key Uses (FDA-Approved Indications):
- Weight loss and maintenance in combination with a reduced-calorie diet and increased physical activity.
- Not for cosmetic weight loss; requires BMI ≥30 (obese) or ≥27 (overweight) with comorbidities.
(Note: The same drug, liraglutide at a lower dose [1.2–1.8 mg], is sold as Victoza for type 2 diabetes management.)
How It Works:
- Appetite suppression: Slows gastric emptying, increases satiety signals in the brain.
- Blood sugar control: Stimulates insulin release, reduces glucagon (evidence from clinical trials like SCALE Obesity and Prediabetes).
- Average weight loss: 5–10% of body weight over 56 weeks (per STEP trials meta-analysis in NEJM, 2021).
Dosage & Administration:
| Week | Dose (subcutaneous injection, once daily) |
|---|---|
| 1 | 0.6 mg |
| 2 | 1.2 mg |
| 3 | 1.8 mg |
| 4 | 2.4 mg |
| 5+ | 3.0 mg (maintenance) |
- Use pre-filled pen; inject into abdomen, thigh, or upper arm.
- Max dose: 3 mg/day. Taper if discontinuing to avoid rebound.
Evidence of Effectiveness:
- SCALE trials (phase 3, >4,800 patients): 63–64% achieved ≥5% weight loss vs. 27–40% placebo (Lancet, 2015).
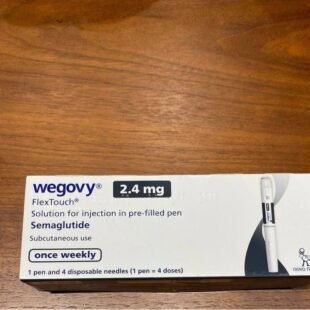
- Sustained results with lifestyle changes; head-to-head vs. semaglutide (Wegovy) shows slightly less efficacy (5–15% vs. 15–20% loss).
Common Side Effects (≥5% incidence):
| Category | Effects |
|---|---|
| GI | Nausea (39%), vomiting (15%), diarrhea (20%), constipation (10%) – often improve over time. |
| Injection | Redness, itching. |
| Other | Headache (14%), fatigue, low blood sugar (if diabetic). |
Serious Risks (Black Box Warning):
- Thyroid C-cell tumors: Rodent studies showed risk; human data limited but monitor for symptoms (lump in neck, hoarseness). Contraindicated in personal/family history of medullary thyroid carcinoma (MTC) or MEN 2.
- Pancreatitis: Acute cases reported (0.3%); stop if suspected.
- Gallbladder disease: Cholecystitis in 1.5% vs. 0.5% placebo.
- Hypoglycemia: Risk with insulin/sulfonylureas.
- Suicidal thoughts: Rare behavioral changes noted.
Contraindications & Warnings:
- Avoid in: MTC/MEN 2 history, severe GI disease, pregnancy (Category C; animal fetal harm).
- Drug interactions: Slows absorption of oral meds (e.g., birth control, antibiotics).
- Cost: ~1,700/month (US, without insurance); patient assistance programs available.
Monitoring & Lifestyle:
- Monthly check-ins for first 16 weeks; discontinue if <4% weight loss.
- Combine with 500 kcal/day deficit diet + 150 min/week exercise for best results (per ADA guidelines).
Sources: FDA label (updated 2023), Novo Nordisk prescribing info, JAMA reviews (2022–2024), clinicaltrials.gov (NCT01272219 et al.)
Additional Information
| Pack | 3 Pens 6mg, ml, 5 Pens 6mg, ml |
|---|